News
Massive Bio Has Onboarded Over 120,000 Cancer Patients to Find Their Clinical Trial
Find Childhood Cancer Clinical Trials
Searching for treatment options in childhood cancer? Massive Bio can help with our free clinical research matching to access innovative therapies for children.
Start Your JourneyCompliant with

You can receive a guidebook with information about childhood cancer by filling out the form.
Clinical Trials for Child Cancer Treatments
Cancer in children can be daunting, but kids with medical support can fight against it. When conventional cancer treatments fade, cancer clinical trials for children become the best alternative. They serve as a new partner in the fight against this disease, bringing hope for enhanced survival rates and minimized side effects. The final objective is to ensure a brighter, healthier future for children.
What is new in pediatric cancer
treatment?
If you are looking for childhood cancer treatment, we can assist you. Our advanced artificial intelligence (AI) platform will recommend options for treatment of pediatric cancers through oncology and hematology clinical trials tailored to your loved one’s condition. Once you submit the patient’s information, our pediatric oncology and hematology experts will review the case and contact you.
Why clinical trials?
Precision Medicine: Tailored treatments, like the ones offered in clinical trials, are based on the genetic footprint of tumors. Identifying specific genetic mutations allows for targeted therapies, potentially reducing side effects and improving outcomes.
Reducing Side Effects: Efforts are ongoing to minimize the long-term side effects of chemotherapy and radiation. Researchers are exploring novel therapies with fewer adverse effects on growing bodies in order to avoid regular chemotherapy.
Collaborative Research: Collaborations among institutions, researchers, and international consortia have accelerated the development of new treatments and clinical trials.
How can Massive Bio help kids with
cancer?
Massive Bio can provide the following services:
Clinical Trial Matching: Our service helps identify innovative pediatric cancer treatments that may be more effective or have fewer side effects than standard options. We specialize in matching pediatric cancer patients with suitable clinical trials.
Access to Experts: The platform connects families with oncology experts who specialize in pediatric cancers. This access ensures that families receive guidance from specialists who understand the nuances and complexities of treating childhood cancers.
Navigating Complexities: Managing this disease can be overwhelming. We support families by providing guidance, resources, and information to navigate the complexities of treatment decisions and available options. If your child is suitable for a clinical trial, we can make this process seamless.
Let’s Learn About Childhood Cancer
Pediatric cancer refers to cancer that develops in children and adolescents, typically under the age of 18. The most common types of pediatric cancers include:
Hematologic cancers:
- Leukemias (cancers of the blood or blood cancers)
- Lymphomas (lymph nodes cancer)
Solid Tumors:
- Brain tumors
- Neuroblastoma (immature nerve cell cancer)
- Osteosarcoma (bone cancer)
- Ewings sarcoma
- Wilms tumor (kidney cancer), among others.
Pediatric cancers can differ from adult cancers. They encompass different types that vary their behavior and response to treatment. Research shows that survival rates for many of these have improved significantly in recent years.
What is an NGS test? Is it important?
NGS stands for Next-Generation Sequencing, a DNA sequencing technology. It quickly reads millions of DNA pieces together, giving a full look at a person’s genes. They provide detailed genetic information about the cancer cells, allowing for more personalized and targeted approaches to therapy.
It can help us identify genetic biomarkers. These are specific genetic mutations within cancer cells that can be targeted and guide treatment decisions. For instance, certain genetic mutations might indicate a better response to a particular therapy or generate the opportunity for creating targeted therapies.
NGS testing is considered part of the standard of care of most cancer types. Discuss this with your child’s oncologist to maximize their possibilities to participate in a clinical trial of interest.
Did you know about ALK-positive solid tumors in
children?
Anaplastic Lymphoma Kinase (ALK) fusion-positive solid tumors can happen in specific types of cancers. The most recognized ALK positive tumors in pediatric patients include:
Rhabdomyosarcoma: Rhabdomyosarcoma is a type of cancer that primarily affects children. It develops from soft tissues, particularly the muscles, and can occur in various parts of the body. This cancer is characterized by the rapid growth of cells that resemble immature muscle cells. Symptoms depend on where the tumor is located, and treatment usually involves a combination of surgery, chemotherapy, and radiation therapy.
Inflammatory Myofibroblastic Tumor (IMT): Inflammatory Myofibroblastic Tumor (IMT) is a rare tumor that can occur in various parts of the body, most commonly in the lungs, abdomen, or pelvis. It’s often considered benign but can behave like a low-grade cancer. IMT is characterized by a mix of inflammatory cells and myofibroblastic cells – cells that are part muscle and part fibrous tissue. The cause of IMT is not well understood, and it can affect people of any age, though it’s more common in children and young adults. Symptoms vary depending on the tumor’s location, and treatment typically involves surgical removal of the tumor.
Neuroblastoma: Is a type of tumor that predominantly affects children and arises from cells of the nervous system. Neuroblastoma primarily affects the adrenal glands but may occur in the abdominal, thoracic, cervical, and pelvic sympathetic ganglia. Its clinical behavior is variable, ranging from spontaneous regression to aggressive metastatic disease.
Glial Tumors (Low and high grade): Are a diverse group of tumors that arise from cells in the nervous system and usually occur in children and young adults. Experts categorize these tumors broadly into low-grade and high-grade based on their aggressiveness.
About treatment for pediatric tumors: standard
vs. novel therapies
The standard treatments typically include surgery, chemotherapy, radiation, immunotherapy, and sometimes stem cell transplants. They aim to eliminate cancer while considering the child’s well-being. Emerging therapies such as immunotherapy and targeted therapies are the forefront of pediatric cancer research.
Clinical trials offer hope for better treatments, improved outcomes, and the advancement of medical knowledge. They provide a pathway to potentially more effective and tailored therapies for children fighting cancer.
By signing up for our matching program, you’re taking an active role in exploring new possibilities and gaining access to an innovative therapy that can potentially transform your life.
We combine the power of technology with our dedicated team of medical providers to find you the best treatment options available.
Access the free matching toolA Childhood Clinical Trial May Be an Option for Your Child
We offer a quick, easy and FREE way to find non-small cell lung cancer clinical research opportunities for patients like you. With our unique clinical trial matching system (CTMS) and team specialized in oncology and hematology, we can rapidly match children to a clinical research study.
Talk to us. Our nurses and patient relations coordinators are here and happy to help you. You are not alone in this fight.
Call +1 844 627 7246You are not alone
You can have an experienced team with you
What Are Clinical Trials?
Cancer is an unfortunate reality that touches most of us at some point in our lives. If you or a loved one has cancer, you may have heard or read that clinical trials could offer access to innovative new treatments. But what exactly is a clinical trial? In this video, Massive Bio co-founder Arturo Loaiza-Bonilla, MD, explains how clinical trials work, what to expect if you enroll in one, and why a clinical trial can be an important treatment option for many cancer patients.
We dream of the day when cancer disappears from our lives. Massive Bio is working tirelessly on achieving that goal.
-
Why should I trust Massive Bio?
Why should I trust Massive Bio?
Massive Bio has provided health to more than 120,000 cancer patients in 25 countries across three continents. It collaborates with over 80 global partners. In 2022, Massive Bio became part of the Precision Cancer Consortium (PCC), alongside major companies such as AstraZeneca, Bayer, Eli Lilly & Company, GSK, Johnson & Johnson/Janssen, Novartis and Roche.
-
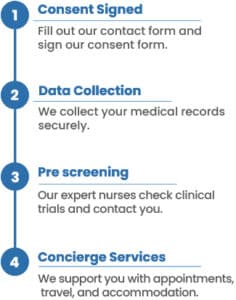
What is the process?
What is the process?
To find the best clinical research studies for you, we need your medical history and consent. You can provide this consent by filling out the form on this page and the following pages. Once you’ve done that, our patient relations coordinator will contact you to discuss the details and provide further information for clinical research matching report.
-
Why do I have to provide my medical records to enroll in a clinical trial?
Why do I have to provide my medical records to enroll in a clinical trial?
To enroll in clinical trial, you must meet highly specific criteria that’s established by the researchers who are conducting the investigation. That includes detailed information about type of cancer, treatment history, response to treatment, and other data that is collected in medical records.
-
What should I do if I don’t have my medical records?
What should I do if I don’t have my medical records?
If you are being treated for cancer or any other disease, your doctor should have a complete record of your medical care, including specific information about what form of the disease you have and what treatments you have received. Your patient relations coordinator will contact you and inform you about the details.
-
What are the costs associated with Massive Bio’s services?
What are the costs associated with Massive Bio’s services?
Massive Bio provides its services to the patients and their doctors at no cost—you won’t have to pay anything to receive a clinical-research matching report. There are no hidden costs involved.
-
How does Massive Bio protect my personal information?
How does Massive Bio protect my personal information?
Massive Bio strictly adheres to all HIPAA guidelines and international regulations focused on maintaining your privacy. We take extra measures to secure your personal information, ensuring it is protected beyond the mandatory requirements.
-
Where can I find clinical research studies in my area?
Where can I find clinical research studies in my area?
Your doctor may know of a clinical research study being conducted in your area that’s recruiting participants and is right for you. However, Massive Bio uses its artificial intelligence-powered platform to match patients to clinical research studies of treatments that give you the best chance of a positive outcome and are being conducted in a geographical location that makes sense for you.
-
Can I continue seeing my doctor or oncologist while also taking services of Massive Bio?
Can I continue seeing my doctor or oncologist while also taking services of Massive Bio?
Yes, Massive Bio keeps your doctor up to date on your status throughout your participation.






