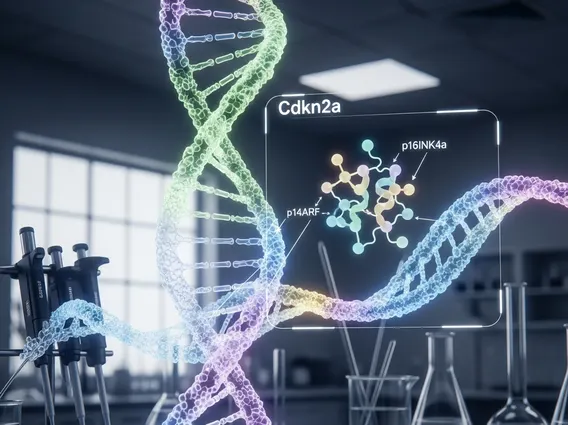
Cdkn2a Gene
The Cdkn2a gene plays a crucial role in cell cycle regulation and tumor suppression. Understanding its function and implications for human health is vital in oncology and genetic research.
Key Takeaways
- The Cdkn2a gene produces proteins p16INK4a and p14ARF, both vital tumor suppressors.
- These proteins regulate cell division and prevent uncontrolled cell growth.
- Mutations in Cdkn2a are strongly linked to an increased risk of various cancers, particularly melanoma and pancreatic cancer.
- Inherited Cdkn2a mutations are a primary cause of familial atypical multiple mole melanoma (FAMMM) syndrome.
- Genetic testing for Cdkn2a mutations can help identify individuals at higher risk, guiding early screening and prevention strategies.
What is Cdkn2a Gene and Its Function?
The Cdkn2a gene (Cyclin-Dependent Kinase Inhibitor 2A) is a critical tumor suppressor gene located on chromosome 9p21. It is unique because it encodes two distinct proteins, p16INK4a and p14ARF (also known as p19ARF in mice), from different reading frames of the same DNA sequence. These proteins act as key regulators of the cell cycle, preventing cells from dividing too rapidly or acquiring cancerous properties. The dual nature of this gene, producing two independent tumor suppressors from a single locus, highlights its profound importance in maintaining genomic stability and preventing oncogenesis.
The primary Cdkn2a gene function involves controlling cell proliferation by interacting with central pathways. The p16INK4a protein specifically inhibits cyclin-dependent kinases (CDK4 and CDK6), which are enzymes that promote cell cycle progression. By blocking these kinases, p16INK4a prevents the cell from moving from the G1 phase to the S phase, effectively pausing cell division and allowing for DNA repair or apoptosis if DNA damage is irreparable. Similarly, the p14ARF protein stabilizes the p53 tumor suppressor protein by inhibiting MDM2, an enzyme that targets p53 for degradation. A stable p53 can then induce cell cycle arrest or apoptosis, further safeguarding against tumor formation. This intricate regulatory network ensures that cells only divide when appropriate, acting as a crucial barrier against uncontrolled growth.
Cdkn2a Gene Mutations and Cancer Risk
Mutations in the Cdkn2a gene are among the most frequently observed genetic alterations in human cancers. These mutations can lead to a loss or reduction in the function of p16INK4a and p14ARF proteins, thereby disrupting the critical cell cycle checkpoints they regulate. Without these functional tumor suppressors, cells can divide uncontrollably, accumulate further genetic damage, and progress towards malignancy. The Cdkn2a gene mutation cancer risk is particularly pronounced for specific cancer types, making it a significant focus in cancer genetics.
One of the most significant associations is the Cdkn2a gene role in melanoma. Inherited mutations in Cdkn2a are the strongest known genetic risk factor for familial melanoma, accounting for a substantial proportion of hereditary cases. Individuals with these germline mutations often develop familial atypical multiple mole melanoma (FAMMM) syndrome, characterized by numerous atypical moles and a significantly increased lifetime risk of melanoma, often at an earlier age. Studies indicate that carriers of Cdkn2a germline mutations have a lifetime risk of melanoma ranging from 50% to 90%, according to the National Cancer Institute. Beyond melanoma, Cdkn2a mutations are also linked to an elevated risk of other cancers, including:
- Pancreatic cancer
- Head and neck squamous cell carcinoma
- Lung cancer
- Brain tumors
Understanding these genetic predispositions allows for targeted screening and surveillance strategies for at-risk individuals, potentially leading to earlier detection and improved outcomes. For instance, individuals with known Cdkn2a mutations may undergo more frequent dermatological examinations and imaging studies. Genetic counseling plays a vital role in helping individuals and families understand their risk, interpret genetic test results, and make informed decisions about preventive measures and early detection protocols. This proactive approach is crucial for managing the elevated cancer risk associated with Cdkn2a mutations.