Carbolic Acid
Carbolic Acid, also known as phenol, is an aromatic organic compound with the chemical formula C6H5OH. Historically significant for its antiseptic properties, its use in modern medicine is highly restricted due to its corrosive and toxic nature.
Key Takeaways
- Carbolic Acid (phenol) is an organic compound historically used as an antiseptic.
- Its Carbolic acid chemical formula is C6H5OH.
- Modern medical uses are limited to specific procedures like chemical peels or sclerotherapy, often in highly diluted forms.
- It poses significant Carbolic acid dangers and safety risks due to its corrosive and toxic properties.
- The History of carbolic acid is closely tied to Joseph Lister’s pioneering work in antiseptic surgery.
What is Carbolic Acid: Definition and Chemical Formula
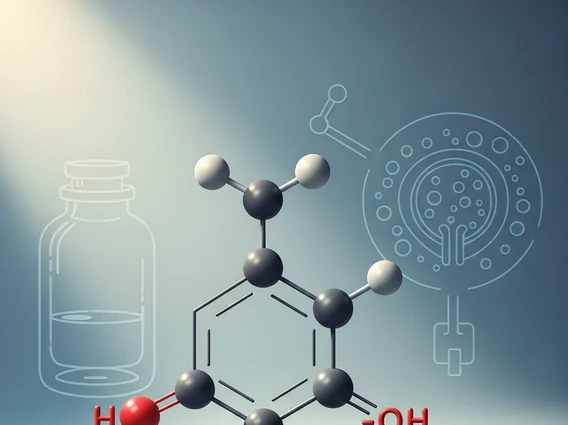
Carbolic Acid, scientifically known as phenol, is a white crystalline solid that is volatile and possesses a distinctive, somewhat sweet, and tarry odor. It is an organic compound characterized by a hydroxyl group (-OH) attached directly to an aromatic hydrocarbon group, specifically a benzene ring. This structure gives it acidic properties, although it is a weak acid. The Carbolic acid chemical formula is C6H5OH, indicating a benzene ring (C6H5) with one hydrogen atom replaced by a hydroxyl group.
In medical and clinical contexts, Carbolic Acid is recognized for its protein-denaturing capabilities, which contribute to its effectiveness as a disinfectant and antiseptic, as well as its caustic effects on tissues. Its solubility in water allows for various concentrations to be prepared, influencing its application and toxicity.
What is Carbolic Acid Used For?
Historically, Carbolic Acid was widely adopted as a groundbreaking antiseptic and disinfectant. Its introduction revolutionized surgical practices by significantly reducing post-operative infections. However, due to its inherent toxicity and the development of safer alternatives, its medical applications have become highly specialized and limited. Today, what is Carbolic Acid used for in clinical settings typically involves controlled, localized applications under strict medical supervision.
Current medical uses include:
- Chemical Peels: Diluted solutions are sometimes used in dermatological procedures for deep chemical peels to treat severe sun damage, wrinkles, or certain skin conditions.
- Sclerosing Agent: In specific cases, it may be used as a sclerosing agent to treat conditions like hemorrhoids or certain cysts, causing tissue to shrink and harden.
- Local Anesthetic: Very dilute solutions can act as a local anesthetic for certain painful conditions, such as ingrown toenails, where its neurolytic properties are utilized to destroy nerve endings.
- Preservative: In extremely low concentrations, it can be found as a preservative in some pharmaceutical preparations and vaccines.
These applications are carefully managed due to the potential for systemic absorption and local tissue damage.
Carbolic Acid Dangers, Safety, and History
The Carbolic acid dangers and safety concerns are significant, primarily due to its corrosive nature and systemic toxicity. Direct contact with skin or mucous membranes can cause severe chemical burns, leading to tissue necrosis. Inhalation of its vapors can irritate the respiratory tract, and ingestion can be fatal, causing damage to the gastrointestinal tract, central nervous system depression, and organ failure. Safe handling requires appropriate personal protective equipment, adequate ventilation, and strict adherence to established safety protocols to prevent exposure.
The History of carbolic acid is notably marked by its pioneering role in antiseptic surgery. In the mid-19th century, British surgeon Joseph Lister observed that carbolic acid could effectively sterilize surgical instruments and wounds, dramatically reducing the high mortality rates from post-operative infections. His work, published in 1867, laid the foundation for modern aseptic surgical techniques. According to historical medical accounts, Lister’s methods using carbolic acid significantly reduced surgical mortality rates from nearly 50% to around 15% in some settings, marking a pivotal moment in medical history. Despite its historical importance, its toxicity led to its gradual replacement by less hazardous and equally effective antiseptics and disinfectants over time.