Isotope
An Isotope, in a medical context, refers to variants of a chemical element that play a crucial role in various diagnostic and therapeutic applications within healthcare. These unique forms of elements are fundamental to understanding and treating numerous conditions, from cancer detection to organ function assessment.

Key Takeaways
- Isotopes are atoms of the same element with an identical number of protons but a differing number of neutrons, leading to variations in atomic mass.
- In medicine, isotopes are primarily used for diagnostic imaging and targeted radiation therapy.
- Radioactive isotopes, or radioisotopes, emit detectable radiation that allows for non-invasive visualization of internal body processes.
- Common examples include Technetium-99m for imaging and Iodine-131 for treating thyroid conditions.
- Their precise application enables early disease detection, accurate staging, and localized treatment delivery.
What is an Isotope?
An Isotope is a variant of a chemical element characterized by having the same number of protons in its atomic nucleus, but a different number of neutrons. This difference in neutron count results in a variation in the atom’s atomic mass, while its chemical properties largely remain consistent due to the identical number of electrons and protons. For instance, all carbon atoms have six protons, but carbon-12 has six neutrons, while carbon-14 has eight neutrons, making them isotopes of carbon.
In medical applications, the focus is often on radioactive isotopes, also known as radioisotopes. These are unstable isotopes that spontaneously decay, emitting radiation in the process. This emitted radiation, which can include alpha particles, beta particles, or gamma rays, is harnessed for both diagnostic imaging and therapeutic interventions. Understanding what are isotopes and how they differ from stable elements is crucial for appreciating their utility in modern medicine.
Examples of Isotopes and Their Applications
The medical field utilizes a diverse range of isotopes, each selected for its specific decay characteristics and biological behavior. These isotopes explained simply, allow clinicians to either visualize internal structures and functions or deliver targeted radiation to diseased tissues. Here are some prominent examples of isotopes and their applications:
- Technetium-99m (Tc-99m): This is one of the most widely used diagnostic radioisotopes. It emits gamma rays, making it ideal for SPECT (Single-Photon Emission Computed Tomography) scans. Tc-99m is employed in bone scans to detect fractures, infections, or tumors; cardiac stress tests to assess blood flow; and kidney scans to evaluate renal function.
- Iodine-131 (I-131): A therapeutic radioisotope primarily used in the treatment of thyroid conditions. The thyroid gland naturally absorbs iodine, allowing I-131 to selectively target and destroy overactive thyroid cells in hyperthyroidism or cancerous thyroid cells in thyroid cancer, minimizing damage to surrounding healthy tissues.
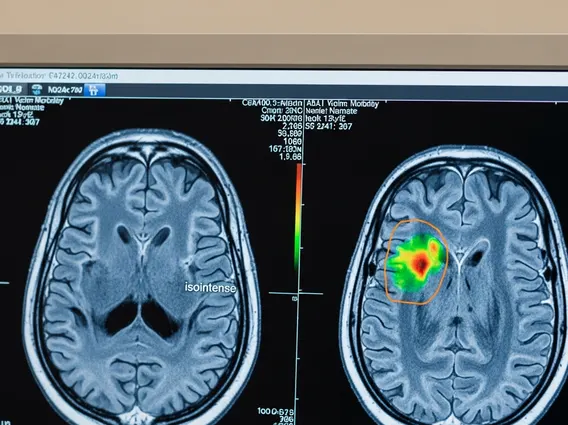
- Fluorine-18 (F-18): Commonly used in Positron Emission Tomography (PET) scans, often incorporated into a glucose molecule to form fluorodeoxyglucose (FDG). Cancer cells typically have a higher metabolic rate and absorb more FDG, making F-18 a powerful tool for detecting and staging various cancers, as well as assessing neurological and cardiac conditions.
- Gallium-67 (Ga-67): Employed in SPECT imaging to detect inflammation, infection, and certain types of tumors, particularly lymphomas. It binds to proteins in inflammatory cells and tumor cells, allowing for their visualization.
How Isotopes Work in Medical Diagnostics and Treatment
The mechanism by which isotopes work in medicine varies depending on their intended application—diagnosis or therapy. In diagnostics, radioisotopes are administered to the patient, usually intravenously, and are designed to concentrate in specific organs, tissues, or pathological areas. As they decay, they emit radiation (typically gamma rays or positrons) that can pass through the body and be detected by specialized external cameras, such as SPECT or PET scanners. These scanners then create detailed images that reveal physiological processes, metabolic activity, or structural abnormalities, providing invaluable information for diagnosis and treatment planning.
For therapeutic purposes, isotopes are chosen for their ability to emit high-energy particles (like beta or alpha particles) that have a short range within biological tissue. These therapeutic radioisotopes are often linked to molecules that specifically target cancer cells or other diseased tissues. Once delivered to the target site, the emitted radiation damages the DNA of the abnormal cells, leading to their destruction while sparing healthy surrounding cells as much as possible. This targeted approach is a cornerstone of nuclear medicine therapy, offering precise and effective treatment options for various cancers and other conditions.