Fluorescein Angiography
Fluorescein Angiography is a diagnostic procedure used to visualize blood flow in the retina and choroid of the eye. It helps ophthalmologists detect and manage various retinal and choroidal conditions by highlighting abnormalities in the eye’s vascular system.
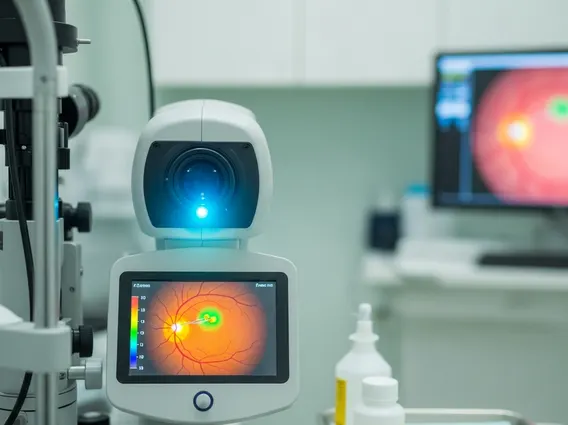
Key Takeaways
- Fluorescein Angiography is an eye test that uses a special dye and camera to examine blood vessels in the retina and choroid.
- It is crucial for diagnosing conditions like diabetic retinopathy, macular degeneration, and retinal vein occlusions.
- The procedure involves injecting a fluorescein dye into a vein, followed by rapid photography of the eye.
- Common side effects are usually mild and temporary, such as nausea or skin discoloration.
- Serious complications are rare but can include allergic reactions.
What is Fluorescein Angiography?
Fluorescein Angiography is a specialized diagnostic test that provides detailed images of the blood vessels in the back of the eye, specifically the retina and choroid. This procedure involves injecting a fluorescent dye, fluorescein, into a vein, typically in the arm. As the dye circulates through the bloodstream and reaches the eye, a series of photographs are taken using a blue light filter, which excites the fluorescein, causing it to glow. This glowing allows ophthalmologists to observe the dye’s flow through the retinal and choroidal vasculature, revealing any blockages, leaks, or abnormal vessel growth. The primary fluorescein angiography purpose is to aid in the diagnosis and management of various eye conditions affecting these delicate structures.
Fluorescein Angiography Procedure Explained
The procedure for Fluorescein Angiography is generally straightforward and takes about 30 minutes to complete. Patients are typically advised to avoid caffeine before the test. The steps involved are:
- Pupil Dilation: Eye drops are administered to dilate the pupils, providing a clearer view of the retina.
- Dye Injection: A small amount of fluorescein dye is injected into a vein, usually in the arm or hand. Patients might experience a brief sensation of warmth or a metallic taste.
- Imaging: As the dye circulates and reaches the eye, a specialized camera takes rapid, sequential photographs of the retina. These images capture the dye’s progression through the blood vessels, highlighting any areas of leakage, non-perfusion (lack of blood flow), or neovascularization (new, abnormal vessel growth).
- Post-Procedure: After the imaging is complete, patients are monitored briefly. The dye will cause the skin to have a yellowish tint and urine to appear bright yellow for up to 24 hours, which is normal.
This diagnostic imaging technique provides invaluable information for conditions such as age-related macular degeneration, diabetic retinopathy, retinal vein occlusions, and various inflammatory eye diseases.
Fluorescein Angiography Side Effects and Risks
While generally safe, Fluorescein Angiography can be associated with certain side effects and risks. Most reactions are mild and temporary. Common side effects include nausea and vomiting, which are the most frequently reported, occurring in approximately 5-10% of patients, usually shortly after the dye injection. Other common, temporary effects include a yellowish discoloration of the skin and eyes, which typically fades within a few hours, and bright yellow urine as the dye is excreted.
More serious side effects are rare but can occur. These include severe allergic reactions (anaphylaxis), which might involve difficulty breathing, swelling, or a significant drop in blood pressure. According to medical literature, severe anaphylactic reactions occur in less than 1 in 1,000 to 1 in 10,000 cases. Other rare risks include fainting, seizures, or cardiac events. Medical staff are always prepared to manage such emergencies. Patients with a history of allergies or kidney problems should inform their doctor before the procedure.