Sotorasib
Sotorasib is a targeted therapy used in the treatment of specific types of cancer, representing a significant advancement in precision oncology. This article explores its function, how it works, its approved uses, and potential adverse effects.
Key Takeaways
- Sotorasib is a targeted cancer therapy specifically designed for certain KRAS G12C-mutated cancers.
- It works by irreversibly binding to the KRAS G12C protein, preventing its activity and inhibiting tumor growth.
- Its primary indication is for adult patients with advanced non-small cell lung cancer (NSCLC) with a KRAS G12C mutation.
- Common side effects include gastrointestinal issues, liver enzyme elevations, and fatigue.
- Regular monitoring is crucial during treatment to manage potential adverse reactions and ensure patient safety.
What is Sotorasib?
Sotorasib is an oral medication classified as a KRAS G12C inhibitor. It represents a significant advancement in precision oncology, specifically targeting cancers driven by a particular genetic mutation. This drug is designed to treat adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) who have a KRAS G12C mutation and have received at least one prior systemic therapy. The development of Sotorasib addressed a long-standing challenge in cancer treatment, as KRAS mutations were previously considered “undruggable” targets. Its approval marked a new era for patients with this specific genetic alteration, offering a targeted approach where conventional therapies might have limited efficacy.
Sotorasib: Mechanism of Action and Clinical Applications
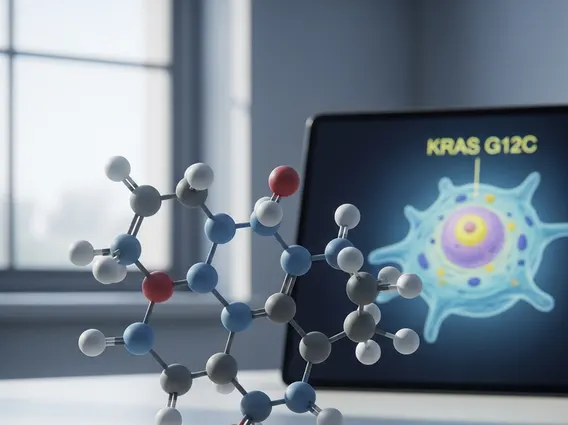
The Sotorasib mechanism of action involves its highly selective and irreversible binding to the cysteine residue of the KRAS G12C mutant protein. By locking KRAS G12C in an inactive state, Sotorasib prevents the protein from signaling downstream pathways that promote cell growth and survival, thereby inhibiting tumor proliferation. This targeted approach minimizes damage to healthy cells, a common issue with traditional chemotherapy.
The primary Sotorasib uses and indications are centered on its efficacy in treating non-small cell lung cancer (NSCLC) with the KRAS G12C mutation. Clinical trials have demonstrated its ability to induce durable responses in patients who have progressed on prior treatments. The FDA granted accelerated approval for Sotorasib based on objective response rates and duration of response observed in these studies. Key indications include:
- Locally advanced or metastatic NSCLC with a KRAS G12C mutation.
- Patients who have received at least one prior systemic therapy.
Ongoing research is exploring its potential in other KRAS G12C-mutated solid tumors, such as colorectal cancer and pancreatic cancer, highlighting its broad therapeutic potential beyond NSCLC.
Potential Side Effects of Sotorasib
Like all medications, Sotorasib can cause adverse reactions. Understanding Sotorasib side effects is crucial for effective patient management. The most frequently reported side effects are generally manageable and include diarrhea, nausea and vomiting, fatigue, musculoskeletal pain, and elevated liver enzymes (ALT/AST).
More serious, though less common, side effects can occur, requiring prompt medical attention. These may include interstitial lung disease/pneumonitis, hepatotoxicity (liver injury), and gastrointestinal toxicities such as severe diarrhea or abdominal pain. Patients undergoing treatment with Sotorasib are closely monitored for these adverse events through regular blood tests and clinical assessments. It is important for patients to report any new or worsening symptoms to their healthcare provider immediately to ensure timely intervention and management.