EGFR Tyrosine Kinase Inhibitor
EGFR Tyrosine Kinase Inhibitors (TKIs) represent a crucial class of targeted therapies in oncology, specifically designed to combat certain cancers by blocking growth-promoting signals. These medications have transformed the treatment landscape for patients with specific genetic mutations.

Key Takeaways
- EGFR TKIs are targeted cancer drugs that inhibit the epidermal growth factor receptor (EGFR) pathway.
- They are primarily indicated for non-small cell lung cancer (NSCLC) patients with activating EGFR mutations.
- These inhibitors work by blocking the tyrosine kinase activity of the EGFR, thereby preventing cancer cell growth and division.
- Different generations of EGFR TKIs exist, offering improved efficacy and strategies to overcome drug resistance.
- Genetic testing for EGFR mutations is essential to identify patients who will benefit from EGFR TKI treatment.
What is an EGFR Tyrosine Kinase Inhibitor (TKI)?
An EGFR Tyrosine Kinase Inhibitor (TKI) is a type of targeted therapy used in cancer treatment, primarily for specific forms of non-small cell lung cancer (NSCLC). These drugs are designed to block the activity of the epidermal growth factor receptor (EGFR), a protein found on the surface of many cells, including cancer cells. When EGFR is overactive or mutated, it can send continuous signals that tell cancer cells to grow and divide uncontrollably. The concept of an EGFR tyrosine kinase inhibitor explained involves understanding that these medications specifically target and interfere with this abnormal signaling pathway.
The effectiveness of EGFR TKIs is highly dependent on the presence of specific activating mutations in the EGFR gene within cancer cells. These mutations make the cancer cells particularly reliant on the EGFR pathway for their survival and proliferation, making them vulnerable to TKI therapy. Identifying these mutations through biomarker testing is a critical step before initiating treatment, ensuring that the therapy is directed at patients most likely to respond.
Mechanism of Action: How EGFR TKIs Work
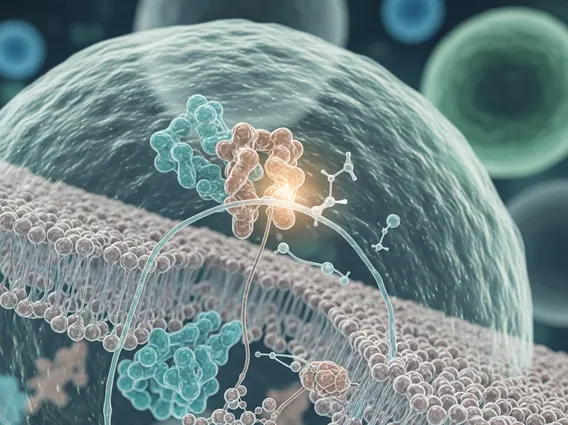
The epidermal growth factor receptor (EGFR) is a protein that spans the cell membrane, with an extracellular domain that binds growth factors and an intracellular domain possessing tyrosine kinase activity. In healthy cells, EGFR activation by its ligands triggers a cascade of intracellular signaling pathways that regulate cell growth, proliferation, differentiation, and survival. However, in many cancers, particularly NSCLC, mutations in the EGFR gene can lead to a constitutively active receptor, meaning it is constantly “on” even without ligand binding. This uncontrolled activation drives tumor growth and progression.
How do EGFR TKIs work mechanism? EGFR TKIs function by competitively binding to the ATP-binding site within the tyrosine kinase domain of the EGFR. By occupying this site, they prevent ATP from binding, which is essential for the phosphorylation of tyrosine residues on the receptor. This inhibition blocks the downstream signaling pathways, such as the RAS/RAF/MEK/ERK and PI3K/AKT/mTOR pathways, which are critical for cell proliferation and survival. Consequently, the cancer cells’ ability to grow, divide, and spread is significantly impaired, leading to tumor regression or stabilization.
EGFR TKI Drugs and Their Clinical Applications
Several generations of EGFR TKI drugs have been developed, each with distinct profiles and indications, primarily for the treatment of advanced non-small cell lung cancer (NSCLC) with activating EGFR mutations. The first-generation EGFR TKIs, such as gefitinib and erlotinib, were groundbreaking in demonstrating the efficacy of targeted therapy for these patients. However, resistance often developed due to secondary mutations, most notably the T790M mutation.
Subsequent generations of EGFR TKIs were designed to overcome these resistance mechanisms. Second-generation drugs like afatinib and dacomitinib irreversibly bind to EGFR, offering broader inhibition. Third-generation EGFR TKIs, such as osimertinib, specifically target the T790M resistance mutation while sparing wild-type EGFR, leading to improved efficacy and reduced side effects. These advancements have significantly improved patient outcomes. EGFR TKI drugs and uses are continually evolving, with ongoing research into new compounds and combination therapies.
Understanding EGFR TKI treatment involves recognizing that these therapies are highly personalized. Patients must undergo molecular testing to confirm the presence of specific EGFR mutations (e.g., exon 19 deletions or L858R point mutations) before treatment initiation. This precision medicine approach ensures that patients receive the most effective therapy for their specific cancer type. While primarily used in NSCLC, research continues to explore the potential role of EGFR TKIs in other cancers where EGFR dysregulation is a driver.